"Guys Google #crisper baby", a young colleague texted the team.
At first, I didn’t realize the misspelling. In the heat of the moment (no pun intended), I thought the term ‘crisper baby’ was referring to some burn-related injury. The fact that I’m a pediatric emergency physician likely informed that pseudo-deduction.
“Not only is the victim a baby, the ‘hashtag crisper baby’ meme is even more unfortunate”, I thought.
Although I wouldn’t put it past social media or tabloids to come up with such cringe-worthy titles, I was intrigued.
Whenever I’m asked by my post-millennial team to Google something, I generally comply. Likely because of my #FOMO (and for the uninitiated in such 3-4 letter acronyms, that stands for ‘Fear Of Missing Out’).
The Googling didn’t take long to make me realize the spelling error. The word was meant to be CRISPR (pronounced “crisper”), alluding to the cutting edge genomic editing technology that had taken the world by storm. The human baby (or babies, twins Lulu and Nana, to be more precise, as I learned later) created in China using CRISPR had created a lot of excitement, and not necessarily the good kind. The births occurred late last year so you may think this is not newsworthy anymore. I believe it is equally important now, given the continuing after effects; just one case in point is it being referred to the ‘CRISPR baby scandal’ by the very prestigious journal Nature.
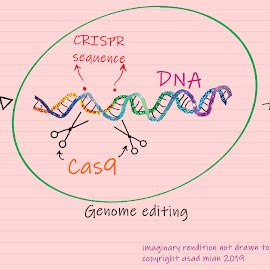
Prior to delving into the ethical debate around it, let me explain what CRISPR is and does. CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats. These are special DNA sequences originally discovered in bacterial genomes. CRISPR works in conjunction with a protein called Cas9 (or ‘CRISPR-associated protein 9’), an enzyme that acts like a pair of molecular scissors that can cut strands of DNA. Taken together, the CRISPR-Cas9 system constitutes a simple, yet powerful editing tool for any genome. It allows researchers to easily alter DNA, which may then lead to modification of gene function. Going into further details of how this system works will be beyond the scope of this blog, but there are excellent credible resources that one can access through the web, if interested in learning further.
Gene editing has great potential in both treating and preventing diseases that are either single gene disorders or those that show more complex inheritance patterns. A few examples of the former for which research is ongoing include cystic fibrosis, hemophilia and sickle cell anemia. From the more complex disease category, there is potential to tackle various cancers, mental illness, heart disease, and HIV infection. The research studies tend to focus on cells and animal models. They have not employed patients in large clinical trials as yet. But this area is opening up. For instance, here’s one scenario: a young woman with a strong family history of breast cancer known to carry a mutation in the BRCA1 gene (linked with breast cancer) opts for CRISPR-Cas9-mediated gene editing through a gene therapy clinical trial. Although this may not be science fact as yet, it may not be too far off in the distant future.
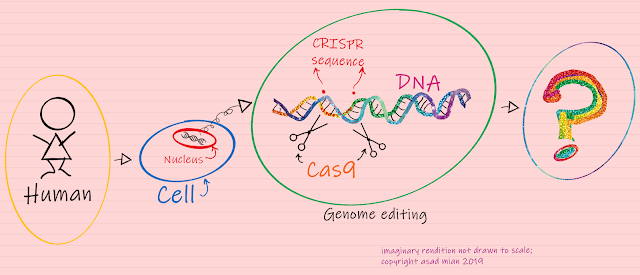 Techniques for modifying genomes have existed for several decades. However, recent innovations around the technology have enabled faster, more efficient and cheaper approaches. Through research, this field of applied biology has been prevalent in non-humans, i.e., ‘model organisms’ such as yeast, bacteria and mice, in the pre-clinical laboratory setting. However, the popular media only tends to take more notice when potential usage in humans is under consideration.
Techniques for modifying genomes have existed for several decades. However, recent innovations around the technology have enabled faster, more efficient and cheaper approaches. Through research, this field of applied biology has been prevalent in non-humans, i.e., ‘model organisms’ such as yeast, bacteria and mice, in the pre-clinical laboratory setting. However, the popular media only tends to take more notice when potential usage in humans is under consideration.
As the science of genome editing gathers momentum, so does the ethics of it. The beneficial aspect of treating or preventing human disease using gene editing in adult somatic (or body) cells may be self-evident. The dilemma arises when germline DNA editing is considered. Germline refers to the reproductive cells, i.e. the sperm and egg. Tinkering of germline DNA through gene editing means that the altered DNA will be transmitted to future generations. The scenario of ‘genomic enhancement’ using CRISPR-Cas9 technology will not be far behind. In other words, parents-to-be who have the money for it, will opt to genetically enhance their babies who on top of zero likelihood of certain genetic disorders, will have enhanced features based on mere parental whims, such as color of skin, height, ‘beauty’ / cosmetic features, etc.
The general consensus of the scientific and non-scientific communities alike is that we are nowhere close to an understanding of the technology and what it can do, to give it a green signal at this stage, either scientifically or ethically. However, what has happened in China with the twins being products of gene-edited germline DNA, we may have to propel this discussion ahead. We need to pause and ponder what are we really getting into?
In the late 90s, as Y2K was approaching, rather than being depressed about the world coming to an end, I was really excited. I was a graduate student in human genetics and at that time the Human Genome Project was in full swing. I was quite full of the young physician-scientist’s chutzpah that complete knowledge of our genetic heritage and subsequent gene therapy successes would be the panacea that the world was seeking. Although I was lured by the love of genes and genomes, there was something lacking towards the end of my graduate studies that precluded my continued work in the field of genetics. I was impatient and I felt that the speed of progress then, using genomics as a tool, was just not fast enough for me.
Now that I’m older and hopefully wiser, I think I tend to be more nuanced in my approach to scientific hype. I used to get energized thinking about genomics, gene therapy, stem cells, and such. A decade or two later, I feel the same is happening with data science, artificial intelligence, machine learning, and the latest craze, genomic editing. CRISPR may be a real game changer per some proponents of the technology. Perhaps they have their own vested interests in extolling this as the next molecular revolution. However, there is still so much to learn and understand about CRISPR and what it can or cannot deliver, that cautious optimism should be the order of the day. Slow and steady, prudent progress in this field will likely go a lot further in pushing the envelope on health innovation for the betterment of humanity the world over.
DISCLAIMER: Copyright belongs to the author. This blog cannot be held responsible for events bearing overt resemblance to any actual occurrences.
Acknowledgment: Author acknowledges Haider Ali and Rayaan Mian for critically reviewing and editing this essay.
About the Author
 Although a pediatrician, ER physician, and researcher by profession, Asad’s proclivity to writing is his means of creative exploration and expression. His articles on health, education, children, humor and popular culture have appeared in newspapers in the US and in Pakistan. Other than the Biloongra series of bilingual books for children, he has authored 'an itinerant observer' a book of short stories/essays published in the US.
Although a pediatrician, ER physician, and researcher by profession, Asad’s proclivity to writing is his means of creative exploration and expression. His articles on health, education, children, humor and popular culture have appeared in newspapers in the US and in Pakistan. Other than the Biloongra series of bilingual books for children, he has authored 'an itinerant observer' a book of short stories/essays published in the US.

